
Rodmar Pulido, President of the Philippine Clinical Research Professionals, recently spoke at the Philippine HIMS BPM Forum about PCRP, and presented the clinical research landscape in the Philippines.
With the huge population of the country, as well as the fact that the majority of healthcare is mainly out of pocket, Pulido said that the Philippines offers sponsors many good patients for clinical trials. Many people have not experienced the drugs taken by individuals in more advanced countries.
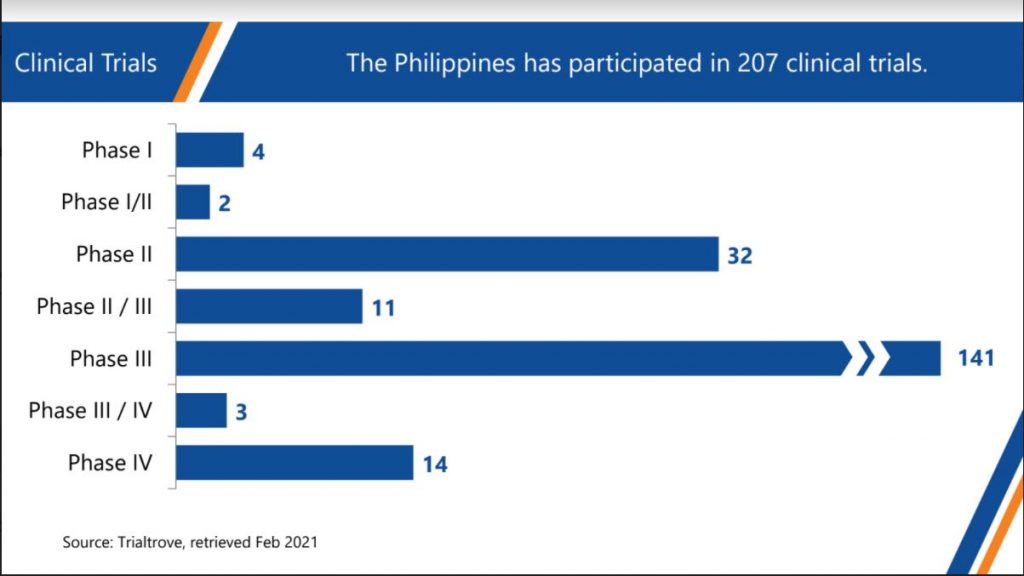
Companies in the Philippines involved in clinical research offer several services. Monitoring, data management, statistics, pharmacovigilance, and logistics are performed for Phase 1, 2 and 3 clinical trials. Phase 4 studies also require many clinical trials, examples of which are real world evidence studies and post marketing surveillance studies.
The Philippines can support clinical trials done outside the country as well. Majority of services performed for overseas trails include data management, statistics, and pharmacovigilance. Because of the Filipino’s proficiency in the English language, projects can be monitored overseas in countries such as Australia and Korea. There are BA/BE (Bioavailability/Bioequivalence) Centers, as well as companies that are involved in pharmacovigilance of drugs, devices, and cosmetics for marketed products. Marketing writing, such as literature reviews, medical evaluation reports, and products of local opportunity studies, is offered too.
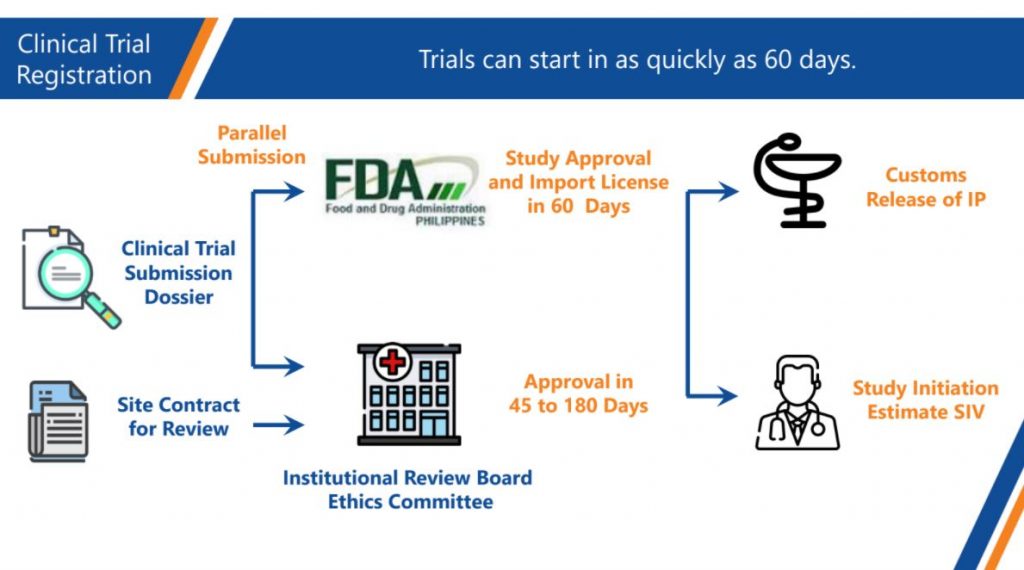
In the Philippines, CROs involved in monitoring clinical trials have to be a licensed by the Philippine FDA. There are thousands of investigators, and 46 hospitals that do clinical trials, with more hospitals for non-interventional trials.
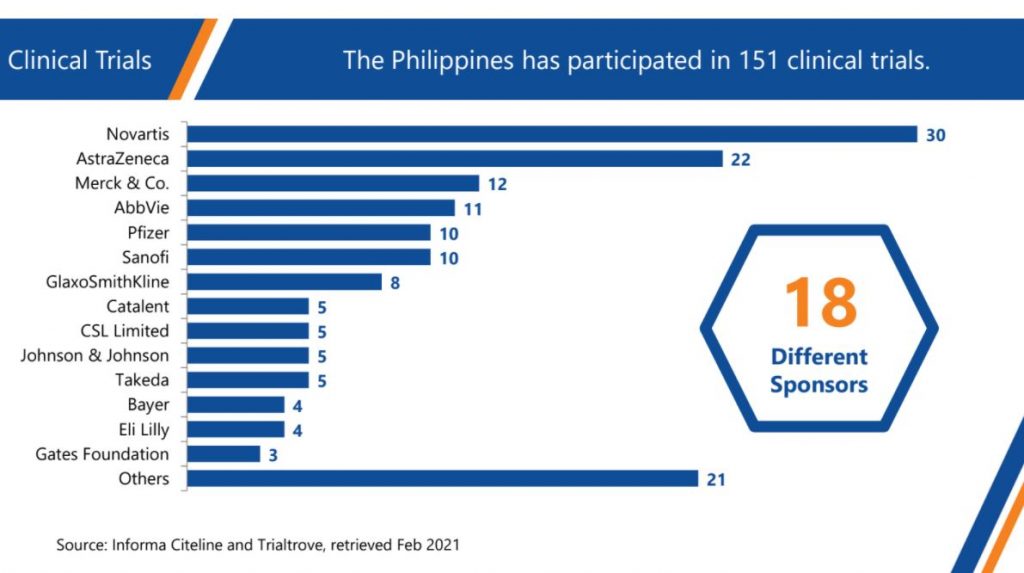
Majority of companies involved in monitoring in the country are multinational companies with affiliates in the Philippines, including the Top 10 globally. The two prominent local CROs are Rainers and PiVOT.
January 27, 2020
March 3, 2020
June 26, 2020
January 14, 2021
February 24, 2021